The initial C-14 level for the calculation can either be estimated, or else directly compared with known year-by-year data from tree-ring data (dendrochronology) to 10,000 years ago, or from cave deposits (speleothems), to about 45,000 years of age. However, it decreases thereafter from radioactive decay, allowing the date of death or fixation to be estimated. Plants fix atmospheric carbon during photosynthesis, so the level of C-14 in plants at the time wood is laid down, or in animals at the time they die, equals the level of C-14 in the atmosphere at that time. One of the frequent uses of the technique is to date organic remains from archaeological sites. In 1960, he was awarded the Nobel Prize in chemistry for this work. Libby estimated that the steady state radioactivity concentration of exchangeable carbon-14 would be about 14 disintegrations per minute (dpm) per gram. The technique was discovered by Willard Libby and his colleagues in 1949 during his tenure as a professor at the University of Chicago. Radiocarbon dating is a radiometric dating method that uses carbon-14 ( 14C) to determine the age of carbonaceous materials up to about 60,000 years old. Radiocarbon dating See main article: Radiocarbon dating Carbon dioxide also dissolves in water and thus permeates the oceans.Ĭarbon-14 can also be produced in ice by fast neutrons causing spallation reactions in oxygen.Ĭarbon-14 then goes through radioactive beta decay.īy emitting an electron and an anti-neutrino, carbon-14 (half life of 5730 years) decays into the stable, non-radioactive isotope nitrogen-14. 
The highest rate of carbon-14 production takes place at altitudes of 9 to 15 km (30,000 to 50,000 feet) and at high geomagnetic latitudes, but the carbon-14 readily mixes and becomes evenly distributed throughout the atmosphere and reacts with oxygen to form radioactive carbon dioxide. The resulting neutrons ( 1n) participate in the following reaction: When cosmic rays enter the atmosphere, they undergo various transformations, including the production of neutrons. Origin and radioactive decay of carbon-14Ĭarbon-14 is produced in the upper layers of the troposphere and the stratosphere by thermal neutrons absorbed by nitrogen atoms. 1 Origin and radioactive decay of carbon-14.This is used in chemical research in a technique called carbon labeling: some carbon-12 atoms of a given compound are replaced with carbon-14 atoms (or some carbon-13 atoms) in order to trace them along chemical reactions involving the given compound. The different isotopes of carbon do not differ appreciably in their chemical properties. The atomic mass of carbon-14 is about 14.003241 amu. The activity of the modern radiocarbon standard is about 14 disintegrations per minute (dpm) per gram carbon. It decays into nitrogen-14 through beta-decay. 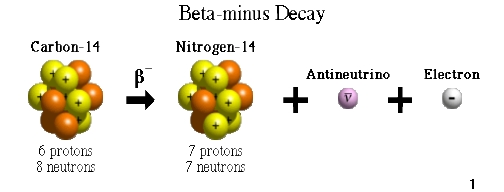
The half-life of carbon-14 is 5730±40 years. There are three naturally occurring isotopes of carbon on Earth: 99% of the carbon is carbon-12, 1% is carbon-13, and carbon-14 occurs in trace amounts, making up as much as 1 part per trillion (0.0000000001%) of the carbon on the Earth. Its presence in organic materials is used extensively as basis of the radiocarbon dating method to date archaeological, geological, and hydrogeological samples. Its nucleus contains 6 protons and 8 neutrons. Carbon-14, 14C, or radiocarbon, is a radioactive isotope of carbon discovered on February 27, 1940, by Martin Kamen and Sam Ruben.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
